Fast pyrolysis holds promise for producing heating and transportation biofuels for the renewable energy market. This article describes how fast pyrolysis works, its advantages and disadvantages, and how it might fit into our renewable energy future.
Table of Contents
- Introduction
- The Fast Pyrolysis Process
- Bio-Oil
- Advantages and Disadvantages
- Summary
- For More Information
- Contributors to This Summary
Introduction
Processing biomass feedstocks such as perennial grasses, woody plants, and agricultural residues into useful fuel products is one of the most critical components of a robust renewable energy system. One promising method of doing so is fast pyrolysis, which converts biomass into liquid biofuel and other energy products. Other ways of producing liquid biofuel include gasification, fermentation, and solvent liquefaction.

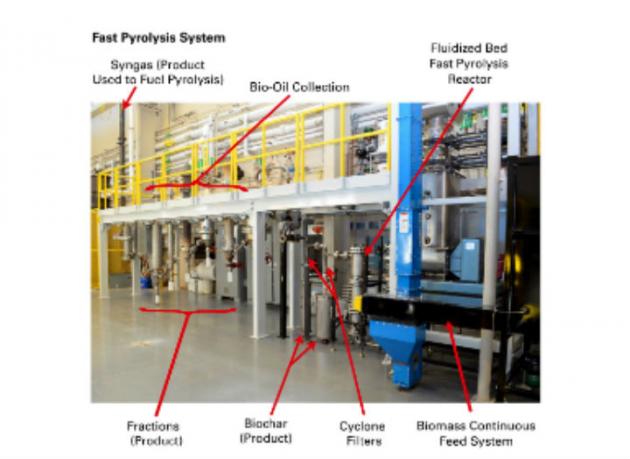
In addition to bio-oil, fast pyrolysis produces a range of other renewable energy products. Although still in its infancy, commercialization of this method is steadily advancing. One approach, shown here in the image, is to build a mobile facility that functions as part of a decentralized system of smaller processing plants that transport bio-oil to a central refining facility.
The Fast Pyrolysis Process
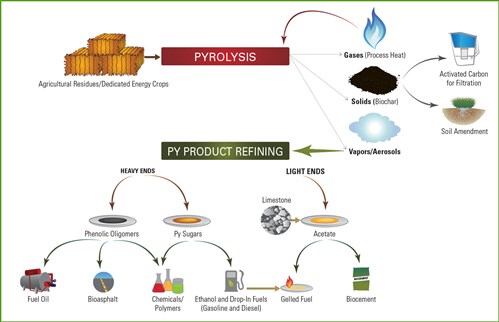
Fast pyrolysis is a thermochemical process during which a lignocellulosic (dry) biomass such as perennial grass, corn stover, or wood is rapidly heated to about 500° C. in the absence of oxygen and then quickly cooled in a reactor. The process converts the biomass into carbohydrate-based compounds that include condensable vapors; these are condensed into liquid bio-oil, the primary product of fast pyrolysis. Fast pyrolysis can convert up to 75 percent of biomass input into bio-oil, yielding about 135 gallons of bio-oil per ton of biomass.
Bio-oil is made up of water and many types of organic compounds that can be easily upgraded into heating oil and, with extensive treatment, transportation fuels. Fast pyrolysis also produces char (charcoal or biochar), and flammable gases and vapors (including methane, hydrogen, and carbon monoxide, which can be burned to heat the reactor thus sustaining the fast pyrolysis process).
Bio-Oil
 Concept for Pyrolysis Refinery Photo courtesy of Bioeconomy Institute at Iowa State University.
Concept for Pyrolysis Refinery Photo courtesy of Bioeconomy Institute at Iowa State University.

Bio-oil is an emulsion of water and organic compounds derived from both the carbohydrate and lignin in the biomass. These organic compounds are mostly alcohols, aldehydes, carboxylic acids, esters, furans, pyrans, ketones, monosaccharides, and anhydrosugars from the carbohydrate and phenolic compounds from the lignin. Through further processing, they can be turned into substitutes for fossil fuel in heating, electricity generation, and transportation. Current estimates are that it would cost between $3 and $4 per gallon to convert corn stover to bio-oil via fast pyrolysis and then upgrade it to gasoline and diesel fuel at a large-capacity refinery.
Bio-oil produced from fast pyrolysis has a low viscosity and is about 15 to 20 percent water. Oxygen is a major contaminant constituent of bio-oil, and contributes to its poor stability in storage or when heated. Adding a catalyst to the pyrolysis process can produce a higher grade of bio-oil through deoxygenation. The resulting bio-oil is easily stored, but in order to be useful must be upgraded into functional products, especially if it will be used as a transportation fuel. Bio-oil can be refined using variations of conventional petroleum refining technology, such as hydrotreating and hydrocracking.
Advantages and Disadvantages of Fast Pyrolysis
Fast pyrolysis has several attractive features:
- The process of making bio-oil is relatively simple and quick.
- All of the biomass components can be processed into a fuel product; none are wasted during the pyrolysis process.
- Pyrolysis reactors are relatively simple and have reached some level of (limited) commercial production.
- A fast pyrolysis facility can be built on a relatively small, mobile scale in order to produce bio-oil close to the biomass source and then transport it to a central facility to be upgraded.
In order to compete with fossil fuels, the products of pyrolysis, in particular bio-oil, must overcome the following hurdles:
- The heating value of bio-oil is only about half that of petroleum-based heating oil by weight.
- Bio-oil is acidic and highly corrosive to metal.
- Bio-oil can contain contaminants.
Summary
The price of fossil fuels has recently dropped precipitously, but no one expects them to stay low forever, nor will our need to reduce greenhouse gas emissions disappear. Therefore it’s essential that we continue to develop all segments of the renewable energy industry to help shrink our consumption of fossil fuels and reduce greenhouse gases.
The commercial future of using fast pyrolysis to convert biomass to energy products is promising. Researchers are steadily developing innovations that increase yields of bio-oil and fractionate the products of fast pyrolysis into compounds that can be more efficiently refined into useable products.
For More Information
- Why Are We Producing Biofuels? Robert C. Brown and Tristan C. Brown. Brownia LLC, Ames, Iowa, 2012.
-
Watch the Bioeconomy Institute’s pyrolysis plant being built! The pilot system contains numerous transducers, thermocouples, sight glasses, and other devices that allow researchers to fully understand the process:
Contributors to This Summary
Authors
-
Robert C. Brown, Anson Marston Distinguished Professor of Engineering and Gary and Donna Hoover Chair in Mechanical Engineering, Iowa State University.
-
Susan Harlow, Freelance Journalist.
Peer Reviewer
-
Samy Sadaka, Assistant Professor, Extension. Biological and Agricultural Engineering, University of Arkansas.
-
Akwasi Boateng, Lead Scientist, USDA-ARS, Eastern Regional Research Center, Wyndmoor, Pennsylvania.
CenUSA Bioenergy is a coordinated research and education effort investigating the creation of a regional system in the Central US for producing advanced transportation fuels from perennial grasses on land that is either unsuitable or marginal for row crop production.  In addition to producing advanced biofuels, the proposed system will improve the sustainability of existing cropping systems by reducing agricultural runoff of nutrients in soil and increasing carbon sequestration.
In addition to producing advanced biofuels, the proposed system will improve the sustainability of existing cropping systems by reducing agricultural runoff of nutrients in soil and increasing carbon sequestration.
CenUSA is supported by Agriculture and Food Research Initiative Competitive Grant no. 2011-68005-30411 from the USDA National Institute of Food and Agriculture.


